Z7_89C21A40L06460A6P4572G3JN0
 Inglés UdeA - Cabezote - WCV(JSR 286)
Inglés UdeA - Cabezote - WCV(JSR 286)
Z7_89C21A40L06460A6P4572G3JQ2
 Signpost
Signpost
Generales
Z7_89C21A40L06460A6P4572G3JQ1
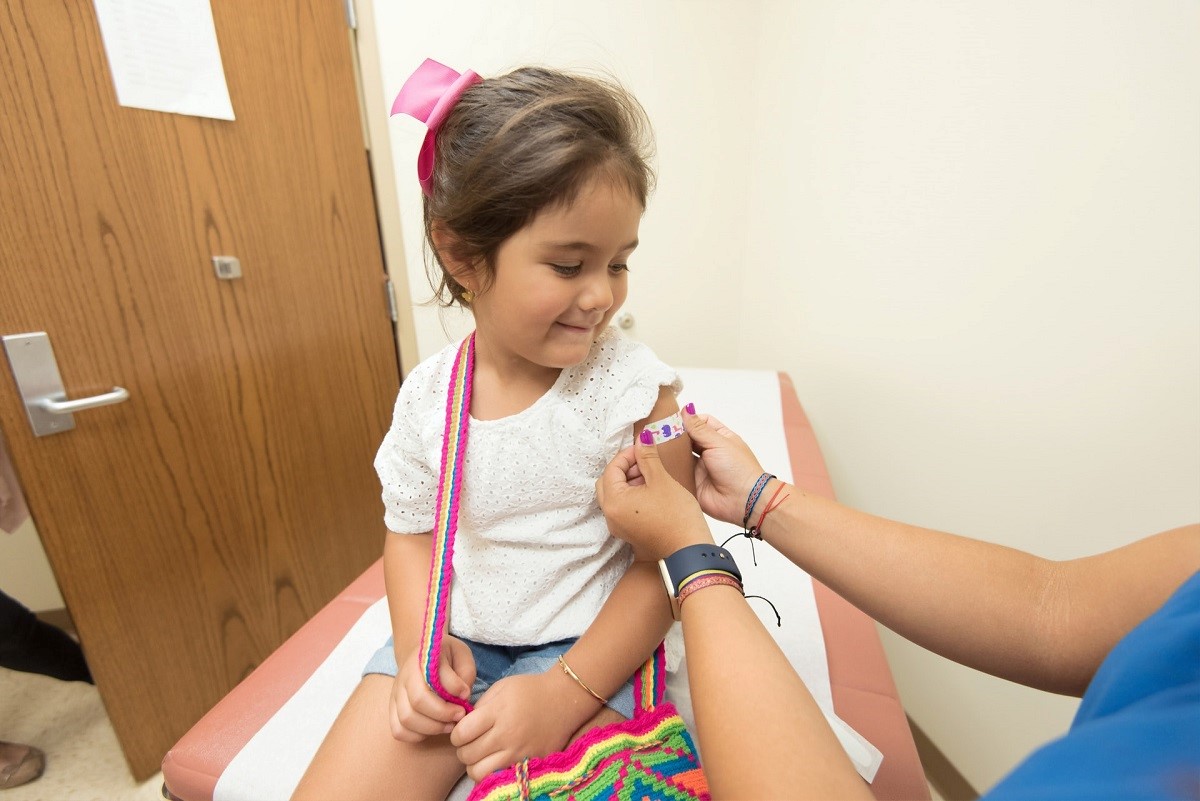
 Effectiveness of Sinovac Vaccine in Children to Be Evaluated by Inmunovirology and PECET
Effectiveness of Sinovac Vaccine in Children to Be Evaluated by Inmunovirology and PECET
Z7_89C21A40L06460A6P4572G3JQ3
 Portal U de A - Redes Sociales - WCV(JSR 286)
Portal U de A - Redes Sociales - WCV(JSR 286)
Z7_89C21A40L0SI60A65EKGKV1K57